Programs
ZIGAKIBART (BION-1301)
Zigakibart is a novel anti-APRIL monoclonal antibody with a potentially disease-modifying approach to treating IgA nephropathy (IgAN) by depleting Gd-IgA1, the pathogenic IgA variant, and reducing proteinuria.
ZIGAKIBART (BION-1301) IN ACTION
Watch our video to learn more about zigakibart’s mechanism of action and disease-modifying potential in IgAN.
Zigakibart is an investigational humanized IgG4 monoclonal antibody that blocks APRIL, a TNF-family cytokine involved in B-cell signaling that is believed to be implicated in IgAN and other indications, from binding to its receptors. Zigakibart is currently enrolling patients in the BEYOND study, a phase 3 clinical trial for patients with IgAN.
A phase 1/2 clinical trial of zigakibart is also currently underway. Parts 1 and 2 of this trial evaluated the safety and tolerability of zigakibart in healthy volunteers, and Part 3 of this trial evaluating zigakibart in patients with IgAN is ongoing.
In healthy volunteers, zigakibart was well-tolerated with no serious adverse events, a pharmacokinetic half-life of approximately 33 days and demonstrated dose-dependent pharmacodynamic effects characterized by durable reductions in serum levels of free APRIL, IgA, galactose-deficient IgA (Gd-IgA1) and IgM, with a lesser reduction in IgG.
In Part 3 of this trial, patients with IgAN in Cohort 1 started treatment at an intravenous, or IV, zigakibart dose of 450 mg every two weeks, and after at least 24 weeks of treatment, all patients transitioned to subcutaneous, or SC, dosing at 600 mg every two weeks. In Cohort 2, patients started treatment at a SC zigakibart dose of 600 mg every two weeks. In both cohorts, patients can receive treatment for up to a total of two years.
Preliminary data from Cohorts 1 and 2 demonstrated that zigakibart has been well-tolerated to date, with no deaths or treatment discontinuations due to adverse events and no anti-drug antibodies were observed. The pharmacokinetics of zigakibart observed in patients with IgAN were consistent with those previously reported in healthy volunteers and sufficient to drive rapid and sustained reductions in free APRIL levels. Zigakibart durably reduced Gd-IgA1, IgA, IgM, and to a lesser extent, IgG levels in patients with IgAN. Zigakibart treatment resulted in proteinuria reductions that were sustained and continued to decline through one year in patients across a range of disease severity.
APRIL
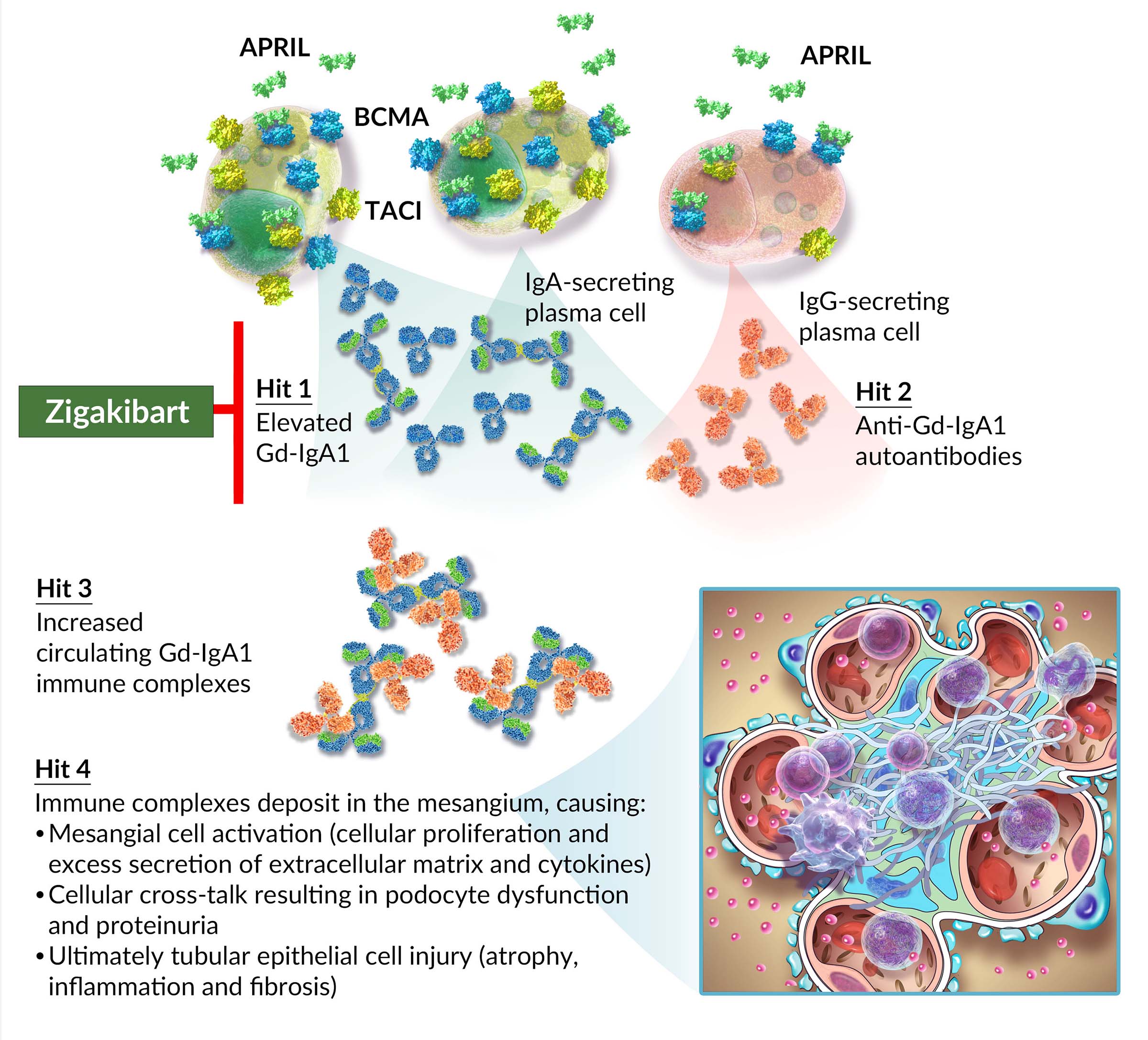
APRIL, which stands for A PRoliferation-Inducing Ligand, is a TNF-family cytokine involved in B-cell signaling, regulating plasma cells and normal levels of immunoglobulin production. Specifically, APRIL binds to BCMA and TACI receptors on the surface of plasma cells to stimulate production of IgA, high levels of which are known to promote the disease process in cancers and autoimmune diseases involving plasma cells.
Patients with IgAN have significantly higher levels of APRIL than healthy individuals, and higher APRIL levels in these patients are correlated with higher Gd-IgA1, proteinuria and lower eGFR. Published literature has demonstrated that APRIL critically drives IgA class switching, the survival of IgA-producing plasma cells and the secretion of Gd-IgA1 (Hit 1 in the multi-hit pathogenesis of IgAN).
Clinical Trials
The BEYOND study is a global, randomized, multicenter, double-blind, placebo-controlled phase 3 clinical trial comparing the safety and efficacy of zigakibart versus placebo in patients with IgAN at risk of progressive loss of kidney function. For more information, visit ClinicalTrials.gov (NCT05852938).
A phase 1/2 clinical trial of zigakibart in healthy volunteers and patients with IgAN is ongoing. Parts 1 and 2 in healthy volunteers have been completed and Part 3 of this trial is enrolling patients with IgAN. For more information, visit ClinicalTrials.gov (NCT03945318).
