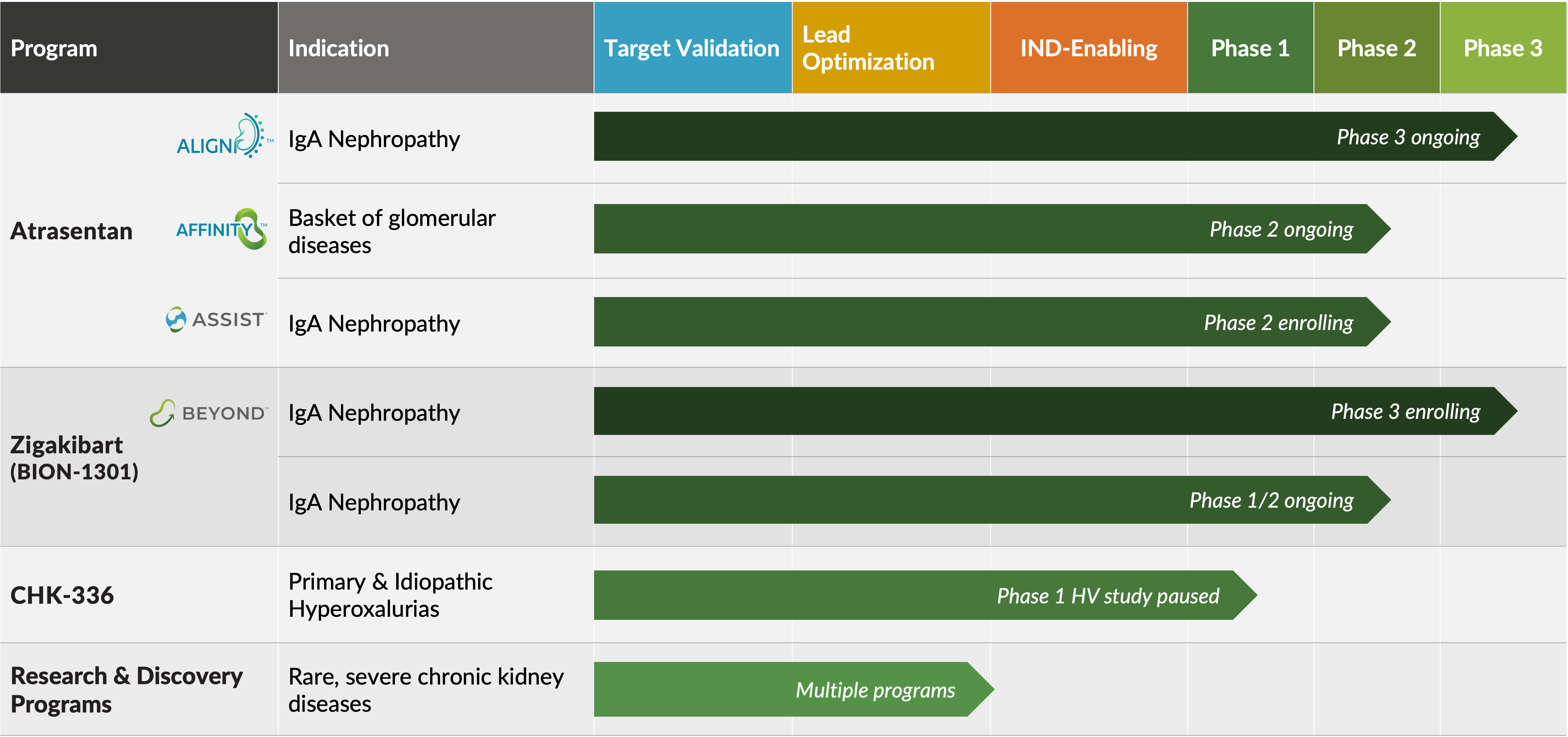
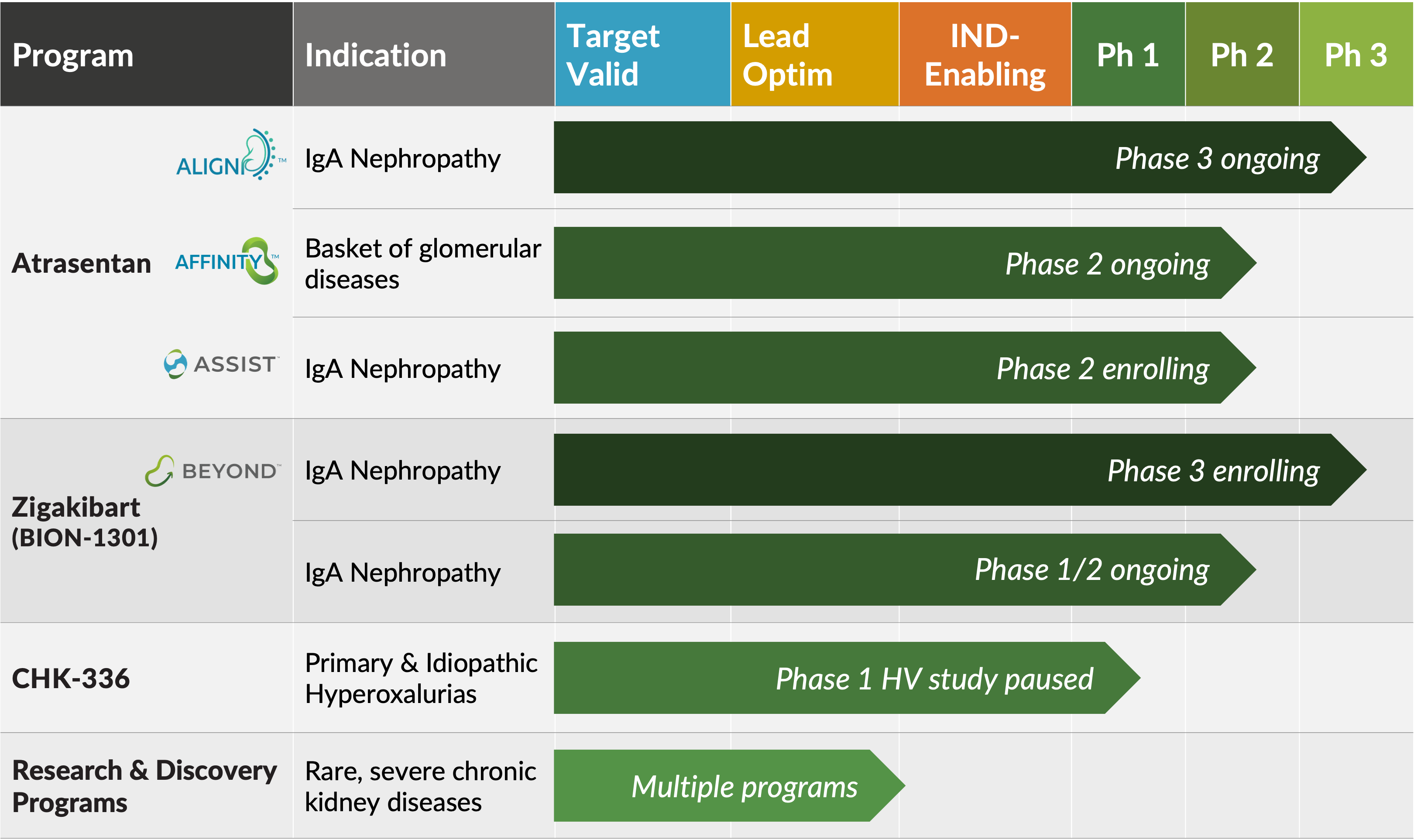
Atrasentan
Atrasentan is a potent and selective endothelin A (ETA) receptor antagonist that is currently being evaluated in a phase 3 registration trial (ALIGN) for IgA nephropathy (IgAN), a phase 2 open-label basket trial (AFFINITY) of proteinuric glomerular diseases including IgAN, FSGS, Alport syndrome and DKD, and a phase 2 crossover trial (ASSIST) for individuals with IgAN on stable doses of a renin-angiotensin system inhibitor (RASi) and an SGLT2 inhibitor (SGLT2i).
Zigakibart (BION-1301)
Zigakibart, a potentially disease-modifying anti-APRIL monoclonal antibody, is being evaluated in a phase 3 trial (BEYOND) and phase 1/2 trial for patients with IgAN.
CHK-336
CHK-336 is an oral small molecule lactate dehydrogenase A inhibitor with liver-targeted tissue distribution for the treatment of primary and idiopathic hyperoxaluria that is currently in phase 1 development.
Research and Discovery
Research and discovery efforts are focused on building a pipeline of precision medicines for rare, severe chronic kidney diseases with defined genetic and molecular drivers.